Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 Zn
Zn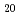
Kitazawa, Takafumi; Ikeda, Yoichi*; Sakakibara, Toshiro*; Matsuo, Akira*; Shimizu, Yusei*; Tokunaga, Yo; Haga, Yoshinori; Kindo, Koichi*; Nambu, Yusuke*; Ikeuchi, Kazuhiko*; et al.
Physical Review B, 108(8), p.085105_1 - 085105_7, 2023/08
Hayashi, Takao; Tobita, Kenji; Nishio, Satoshi; Ikeda, Kazuki*; Nakamori, Yuko*; Orimo, Shinichi*; DEMO Plant Design Team
Fusion Engineering and Design, 81(8-14), p.1285 - 1290, 2006/02
Times Cited Count:22 Percentile:79.98(Nuclear Science & Technology)Neutron transport calculations were carried out to evaluate the capability of metal hydrides and borohydrides as an advanced shielding material. Some hydrides indicated considerably higher hydrogen content than polyethylene and solid hydrogen. The hydrogen-rich hydrides show superior neutron shielding capability to the conventional materials. From the temperature dependence of dissociation pressure, ZrH and TiH
and TiH can be used without releasing hydrogen at the temperature of less than 640
can be used without releasing hydrogen at the temperature of less than 640  C at 1 atm. ZrH
C at 1 atm. ZrH and Mg(BH
and Mg(BH )
) can reduce the thickness of the shield by 30% and 20% compared to a combination of steel and water, respectively. Mixing some hydrides with F82H produces considerable effects in
can reduce the thickness of the shield by 30% and 20% compared to a combination of steel and water, respectively. Mixing some hydrides with F82H produces considerable effects in  -ray shielding. The neutron and
-ray shielding. The neutron and  -ray shielding capabilities decrease in order of ZrH
-ray shielding capabilities decrease in order of ZrH
 Mg(BH
Mg(BH )
) and F82H
and F82H  TiH
TiH and F82H
and F82H  water and F82H.
water and F82H.
 system
systemKurosaki, Yuzuru; Takayanagi, Toshiyuki*
Chemical Physics Letters, 406(1-3), p.121 - 125, 2005/04
no abstracts in English
Uchida, Shunsuke*; Sato, Tomonori; Morishima, Yusuke*; Hirose, Tatsuya*; Miyazawa, Takahiro*; Kakinuma, Nagao*; Sato, Yoshiyuki*; Usui, Naoshi*; Wada, Yoichi*
Proceedings of 12th International Conference on Environmental Degradation of Materials in Nuclear Power Systems-Water Reactors (CD-ROM), p.19 - 29, 2005/00
Static and dynamic responses of stainless steel specimens exposed to H O
O and O
and O in high temperature water were evaluated by analyzing ECP and FDCI (frequency dependent complex impedance). The oxide films on the specimens were characterized by multilateral surface analyses, e.g., LRS, SIMS, XPS and direct electric resistance measurement. As a result of evaluation, it was confirmed that (1) corrosive condition of BWR normal water chemistry (NWC) was simulated by 100 ppb H
in high temperature water were evaluated by analyzing ECP and FDCI (frequency dependent complex impedance). The oxide films on the specimens were characterized by multilateral surface analyses, e.g., LRS, SIMS, XPS and direct electric resistance measurement. As a result of evaluation, it was confirmed that (1) corrosive condition of BWR normal water chemistry (NWC) was simulated by 100 ppb H O
O without co-existing O
without co-existing O , while that of hydrogen water chemistry (HWC) was simulated by 10 ppb H
, while that of hydrogen water chemistry (HWC) was simulated by 10 ppb H O
O , (2) ECP under HWC was as high as that under NWC, while dissolution rate of oxide film under HWC was much lower than that under NWC, (3) combination effects of electric resistance and dissolution rate of oxide caused same level ECP for both NWC and HWC, and (4) distinct weight loss of the specimen exposed to 100 ppb H
, (2) ECP under HWC was as high as that under NWC, while dissolution rate of oxide film under HWC was much lower than that under NWC, (3) combination effects of electric resistance and dissolution rate of oxide caused same level ECP for both NWC and HWC, and (4) distinct weight loss of the specimen exposed to 100 ppb H O
O was observed.
was observed.
 potential energy surfaces for the lowest three doublet states (1
potential energy surfaces for the lowest three doublet states (1 A', 2
A', 2 A', and 1
A', and 1 A") of the BrH
A") of the BrH system
systemKurosaki, Yuzuru; Takayanagi, Toshiyuki
Journal of Chemical Physics, 119(15), p.7838 - 7856, 2003/10
Times Cited Count:26 Percentile:63.62(Chemistry, Physical)Adiabatic potential energy surfaces of the lowest three doublet states (1 A', 2
A', 2 A', and 1
A', and 1 A") for the BrH
A") for the BrH system have been calculated globally using the MRCI+Q method with the aug-cc-pVTZ basis set. Spin-orbit effects were considered on the basis of Breit-Pauli Hamiltonian. The calculated adiabatic energies were fitted to the analytical functional form of many-body expansion. The barrier heights of the abstraction and exchange reactions on the ground-state PES were calculated to be 1.28 and 11.71 kcal mol
system have been calculated globally using the MRCI+Q method with the aug-cc-pVTZ basis set. Spin-orbit effects were considered on the basis of Breit-Pauli Hamiltonian. The calculated adiabatic energies were fitted to the analytical functional form of many-body expansion. The barrier heights of the abstraction and exchange reactions on the ground-state PES were calculated to be 1.28 and 11.71 kcal mol , respectively, at the MRCI+Q/aug-cc-pVTZ level of theory. The fits for the three PESs were successful within the accuracy of 0.1 kcal mol
, respectively, at the MRCI+Q/aug-cc-pVTZ level of theory. The fits for the three PESs were successful within the accuracy of 0.1 kcal mol . Thermal rate constants for the abstraction and exchange reactions and their isotopic variants were calculated with the fitted 1
. Thermal rate constants for the abstraction and exchange reactions and their isotopic variants were calculated with the fitted 1 A' PES using the ICVT/LAG method. The calculated rate constants for the abstarction reactions agree fairly well with experiment but those for the exchange reactions were much smaller than experiment, which suggests that the reliable experimental data are still insufficient.
A' PES using the ICVT/LAG method. The calculated rate constants for the abstarction reactions agree fairly well with experiment but those for the exchange reactions were much smaller than experiment, which suggests that the reliable experimental data are still insufficient.
 O
OKameda, Yasuo*; Sasaki, Motoya*; Usuki, Takeshi*; Otomo, Toshiya*; Ito, Keiji*; Suzuya, Kentaro; Fukunaga, Toshiharu*
Journal of Neutron Research, 11(3), p.153 - 163, 2003/09
We describe results of time-of-flight (TOF) neutron diffraction of the liquid water null-H O in order to investigate the effect of both the scattering angle and the neutron flight path ratio to the observed self-scattering intensities. An empirical inelasticity correction procedure is proposed using the self-scattering intensity observed for the null-H
O in order to investigate the effect of both the scattering angle and the neutron flight path ratio to the observed self-scattering intensities. An empirical inelasticity correction procedure is proposed using the self-scattering intensity observed for the null-H O.
O.
 Si
Si under pressure
under pressureAraki, Shingo; Nakashima, Miho*; Settai, Rikio*; Kobayashi, Tatsuo*; Onuki, Yoshichika
Acta Physica Polonica B, 34(2, Part1), p.439 - 442, 2003/02
no abstracts in English
Seguchi, Tadao
Nuclear Instruments and Methods in Physics Research B, 185(1-4), p.43 - 49, 2001/12
Times Cited Count:18 Percentile:48.84(Instruments & Instrumentation)no abstracts in English
 , and H
, and H O relevant to edge plasma impurities
O relevant to edge plasma impuritiesShirai, Toshizo; Tabata, Tatsuo*; Tawara, Hiroyuki*
Atomic Data and Nuclear Data Tables, 79(1), p.143 - 184, 2001/09
Times Cited Count:56 Percentile:91.21(Physics, Atomic, Molecular & Chemical)no abstracts in English
 /He mixture gas
/He mixture gasTanzawa, Sadamitsu; Hiroki, Seiji; Abe, Tetsuya; Inohara, Takashi*
Shinku, 44(7), p.667 - 670, 2001/07
no abstracts in English
Teshigawara, Makoto; Meigo, Shinichiro; Sakata, Hideaki*; Kai, Tetsuya; Harada, Masahide; Ikeda, Yujiro; Watanabe, Noboru
JAERI-Research 2001-022, 33 Pages, 2001/05
no abstracts in English
 +H
+H collisions calculated with the trajectory-surface-hopping method
collisions calculated with the trajectory-surface-hopping methodIchihara, Akira; Iwamoto, Osamu; Yokoyama, Keiichi
Atomic and Plasma-Material Interaction Data for Fusion, Vol. 9, p.193 - 235, 2001/00
no abstracts in English
 +H
+H (v=0-14)
(v=0-14) H+H
H+H at low collision energies
at low collision energiesIchihara, Akira; Iwamoto, Osamu; Janev, R. K.*
Journal of Physics B; Atomic, Molecular and Optical Physics, 33(21), p.4747 - 4758, 2000/11
Times Cited Count:62 Percentile:90.25(Optics)no abstracts in English
 , H
, H , H
, H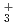 , H, H
, H, H and H
and H with hydrogen molecules
with hydrogen moleculesTabata, Tatsuo*; Shirai, Toshizo
Atomic Data and Nuclear Data Tables, 76(1), p.1 - 25, 2000/09
Times Cited Count:79 Percentile:94.46(Physics, Atomic, Molecular & Chemical)no abstracts in English
Sakurai, Tsutomu*; Yokoyama, Atsushi
Journal of Nuclear Science and Technology, 37(9), p.814 - 820, 2000/09
no abstracts in English
Sekiguchi, Hiromi*; Sekiguchi, Tetsuhiro; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Shimada, Hiromichi*; Baba, Yuji
Surface Science, 454-456, p.407 - 411, 2000/05
Times Cited Count:5 Percentile:32.74(Chemistry, Physical)no abstracts in English
 O+SO
O+SO atmosphere
atmosphereKurata, Yuji; Suzuki, Tomio; Shimizu, Saburo
JAERI-Research 2000-011, p.56 - 0, 2000/03
no abstracts in English
 Si
Si and response to hydrostatic pressure as studied by neutron diffraction
and response to hydrostatic pressure as studied by neutron diffractionKawarazaki, Shuzo*; Sato, Masugu*; Miyako, Yoshihito*; Chigusa, Nobusato*; Watanabe, Kenji*; Metoki, Naoto; Koike, Yoshihiro; Nishi, Masakazu*
Physical Review B, 61(6), p.4167 - 4173, 2000/02
Times Cited Count:86 Percentile:93.71(Materials Science, Multidisciplinary)no abstracts in English
 +H
+H collisions
collisionsIchihara, Akira; Iwamoto, Osamu; Yokoyama, Keiichi
Atomic Collision Research in Japan, No.25, p.28 - 29, 1999/11
no abstracts in English
Sekiguchi, Hiromi*; Sekiguchi, Tetsuhiro; Imamura, Motoyasu*; Matsubayashi, Nobuyuki*; Shimada, Hiromichi*; Baba, Yuji
Photon Factory Activity Report 1998, Part B, P. 37, 1999/11
no abstracts in English